Alpha GPC 50%/99%
Alpha GPC 50%/99% available to ship from USA with third party test reports, guaranteed quality that exceeds your requirement.
Click the button below to get a free quote today!
The Benefits of Alpha GPC
Enhanced Focus
Alpha GPC enhances focus leading to better productivity.
Improved Memory
Alpha GPC improves functions of the brain's neurotransmitters.
Boosts Learning
Alpha GPC boosts mood creating a better ambiance.
Increased BRAIN OXYGENATION
Alpha GPC helps in increases people's brain oxygenation.
Improved Athletic Performance
Alpha GPC increases strenght leading to better performance.
Increases physical endurance
Alpha GPC aids in improving recovery of the brain after an accident.
USA Fresh Stock
What is Alpha GPC ?

Alpha GPC or Alpha-glycerophosphocholine (aGPC), is a choline-containing phospholipid isolated from soy. A wealth of research on this compound supports a role for enhancing cognitive function, increasing strength and stimulating the release of growth hormone.
Other uses for Alpha-GPC include improment of various kinds of dementia, stroke, and “mini-stroke” (transient ischemic attack, TIA). Alpha-GPC is also used for improving memory, thinking skills, and learning.

Alpha GPC
Our Advantage
Best quality ingredient Alpha GPC from Nutri Avenue Inc
USA fresh stock
We have prepared sufficient inventory in our warehouse in California USA which provides you stable supply on ingredient demands.
3rd party test report
We will do strict test on each batch of Alpha GPC to ensure that the purity of each batch can meet your request.
10% below market price
Through our mass production, we are able to reduce Alpha GPC production costs, reducing procurement costs for our customers by at least 10%.
Free samples
We can provide free samples from our warehouses in USA and China for your inspection before the order.

SCIENCE
L-Alpha glycerylphosphorylcholine (alpha-GPC, choline alfoscerate) is a natural choline compound found in the brain. It is also a parasympathomimetic acetylcholine precursor which has been investigated for its potential for the treatment of Alzheimer’s disease[2] and other dementias.
Alpha-GPC rapidly delivers choline to the brain across the blood–brain barrier and is a biosynthetic precursor of acetylcholine. It is a non-prescription drug in most countries. The FDA determined that intake of no more than 196.2 mg/person/day is considered generally recognized as safe.
RESEARCH
An Italian multicentre clinical trial on 2,044 patients suffering from recent stroke were supplied alpha-GPC in doses of 1,000 mg/day for 28 days and 400 mg three times per day for the five ensuing months. The trial confirmed the therapeutic role of alpha-GPC on the cognitive recovery of patients based on four measurement scales (Mathew Scale (MS), Mini Mental State Test (MMST), Crichton Rating Scale (CRS) and the Global Deterioration Scale (GDS)) three of which reached statistical significance.[non-primary source needed] In trials utilizing alpha-GPC in vascular dementia, alpha-GPC administration was reported to improve performance on psychometric tests and to be well tolerated.
Small scale studies focusing on the effects of alpha-GPC on physical performance have also reported that alpha-GPC supplementation can increase maximum power and velocity in specified tests (counter-movement jump test) and increase lower body force (isometric mid-thigh pull test). In iron deficient women, alpha-GPC supplementation has also been reported to enhance non-heme iron uptake from dietary sources.
PRODUCTION
Industrially, alpha-GPC is produced by the chemical or enzymatic deacylation of phosphatidylcholine enriched soya phospholipids followed by chromatographic purification. Alpha-GPC may also be derived in small amounts from highly purified soy lecithin as well from purified sunflower lecithin.

L-Alpha-glycerylphosphorylcholine (GPC) is a widely used food supplement. GPC has been shown to exert beneficial effects in several organs; however, the cardiac effects of GPC have yet to be investigated. The aim of the present study was therefore to map out the effects of GPC on cardiac myocytes, with or without ischemia–reperfusion insult. Neonatal rat cardiac myocytes were treated with GPC at 1, 10, 80, and 100 µM concentrations for 15 min, 3 h, or 24 h, respectively. Cell viability by calcein assay and the degree of oxidative stress by DHE (superoxide level) and H2DCF (total ROS accumulation) staining were measured. In separate experiments, cardiomyocytes were pre-treated with the optimal concentration of GPC for 3 h and then cells were exposed to 4 h of simulated ischemia followed by 2 h of reperfusion (SI/R). Cell viability was measured at the end of the SI/R protocol. In normoxic conditions, the 15-min and the 3-h GPC treatment did not affect cell viability, total ROS, and superoxide levels. Under SI/R conditions, the 3-h GPC treatment protected the cardiac myocytes from SI/R-induced cell death and did not alter the level of oxidative stress. The 24-h GPC treatment in normoxic conditions resulted in significant cell death and increased oxidative stress at each concentration. Here we provide the first evidence for the cytoprotective effect of short-term GPC treatment. However, long-term administration of GPC may exert cytotoxicity in a wide concentration range in cardiac myocytes. These results may draw attention to a comprehensive cardiac safety protocol for the testing of GPC.
INTRODUCTION
L-Alpha-glycerylphosphorylcholine (choline alphoscerate, GPC) is a natural endogenously produced choline derivative and acetylcholine precursor in the brain which (in the form of a synthetic compound) is widely used as a food supplement. After oral ingestion, it is converted metabolically to phosphatidylcholine, the active form of choline that is able to increase acetylcholine levels in the brain . In terms of its use as a food/health supplement, oral GPC intake/supplementation has gained growing attention in both the media and in scientific areas. Since 1998, the Institute of Medicine of the National Academy of Sciences (USA) has recognized choline as an essential nutrient for humans and has made recommendations for the dietary choline intake (Institute of Medicine and National Academy of Sciences 1998). It has been shown that GPC supplements may help substitute for insufficient dietary choline . GPC also contributes to the synthesis of membrane phospho- and glycerolipids, and thus positively influences membrane fluidity.
GPC is generally considered a safe and non-toxic compound and it is classified as GRAS (Generally recognized as safe) in the USA (GRN 000419). In Italy and Poland, GPC is a registered drug (Anatomical Therapeutic Chemical Classification System code: n07ax02) marketed under the trade name Gliatilin (produced by Italfarmaco S.p.A., Milan; Italy). It is recommended for use in humans to support brain health and mental capacity. A multicentric, clinical trial confirmed the therapeutic role of alpha-GPC on the cognitive recovery of patients with acute stroke or transient ischemic attack . Along this line, GPC was shown to exert neuroprotective effects in some mental disorders in a series of human clinical studies .
In addition to its neuroprotective effects, GPC was documented to preserve mitochondrial respiration in liver mitochondria and to reduce hepatic ischemia-induced oxidative stress and inflammation in rodent models of ischemia/reperfusion (I/R) . Protective effects of GPC associated with reduced radical production have been documented also in a relevant rodent model of mesenteric I/R . In view of this, we hypothesized that GPC could prevent oxidative damage following I/R insult in other tissues, including the myocardium.
One concern of GPC is determining the correct dosage, as it is reported that inadequate intake or abnormal metabolism of choline is associated with the increased risk of cardiovascular disease and various cancers . Dose–response experiments were so far restricted to the brain. To the best of our knowledge, the cardiac effects of GPC have yet to be examined.
Therefore, the major objective of the present study was to evaluate the timing and doses of GPC treatment on isolated cultured cardiac myocytes, with a special focus on observing the effects of GPC on the degree of oxidative stress. We aimed to test the hypothesis that GPC could prevent ischemia-induced cell death and oxidative stress in cardiac myocytes subjected to simulated I/R.
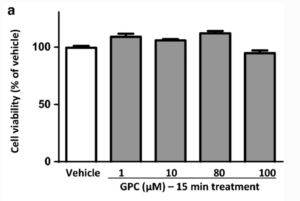
RESULTS
Acute effect of GPC in isolated NRCMs in normoxic conditions
In normoxic conditions, the acute (15 min) treatment with different concentrations of GPC had no impact on the cell viability of NRCMs in comparison to the vehicle control (Fig. 2a). Neither the extent of superoxide production nor total ROS accumulation changed significantly after 15-min exposure to GCP in cardiac myocytes (Fig. 2b, c). On the basis of our earlier findings by Strifler and co-workers, acute effects of GPC on mitochondrial respiration were also investigated in this setup, and interestingly, treatment with 80–100 µM GPC heavily increased the oxygen consumption rate (OCR) of cardiac myocytes (Supplementary Fig. 2).
There are some limitations in the present study as this is the first study to investigate the effects of a broad concentration range of GPC on cardiac cells. Nevertheless, in vivo experiments would certainly supply further information regarding the functional cardiac effects of GPC. The mechanisms of action of GPC on cardiac cells are poorly characterized, and therefore it should be studied with multiple omics technologies in any depth.
In conclusion, in the present study, we have shown for the first time that choline donor L-Alpha-GPC (which is widely used as a food supplement and generally considered as safe for human use) had ambiguous effects on cardiac cells. It may be beneficial in short-term administration to maintain the physiological balance of ROS production under normoxic, healthy conditions and could be also protective in I/R conditions, but could, in fact, be cytotoxic if it surrounded the cells for long enough. Besides the duration of the treatment, the correct dosage can also be a crucial factor, as a fine-tuning effect seemed to occur in a small, but dietary-relevant concentration range. Thus (despite many limitations of this in vitro study), our results indicate the need for a comprehensive cardiac safety testing of GPC.
| Chemical Name: | Alpha GPC 50% |
| CAS Number: | 28319-77-9 |
| Molecular Formula: | C8H20NO6P |
| Molecular Weight: | 257.22.mol |
| Packaging: | In paper-drums and two layers of poly bags inside. Net weight: 25kg/drum |
| Main Benefits: | Used for Focus, Memory, Cognitive Ability, and Growth Hormone Support |
| Application: | Dietary Supplements |
Dosage Notes
Alpha GPC is about 40% choline by weight. So 1,000 mg of Alpha GPC provides approximately 400 mg of choline.
- Alpha GPC suggested dosage for cognitive benefits is 400 – 1,200 mg per day.
- Athletic training suggested dosage of Alpha GPC is 400 mg first thing in the morning, and another 400 mg dose 15 – 30 minutes before working out.
- Clinical treatment of Alzheimer’s Disease, dementia and other cognitive disorders dosage of up to 1,200 mg per day.
For higher dosages, split the daily Alpha GPC total into 2 or 3 doses per day. For example, 1,200 mg would be taken 400 mg at a time.
References
- ^ De Jesus Moreno Moreno M (January 2003). “Cognitive improvement in mild to moderate Alzheimer’s dementia after treatment with the acetylcholine precursor choline alfoscerate: a multicenter, double-blind, randomized, placebo-controlled trial”. Clinical Therapeutics. 25 (1): 178–93. doi:10.1016/S0149-2918(03)90023-3. PMID 12637119.
- ^ Jump up to:a b Parnetti L, Mignini F, Tomassoni D, Traini E, Amenta F (June 2007). “Cholinergic precursors in the treatment of cognitive impairment of vascular origin: ineffective approaches or need for re-evaluation?”. Journal of the Neurological Sciences. 257 (1–2): 264–9. doi:10.1016/j.jns.2007.01.043. PMID 17331541. S2CID 34661218.
- ^ Doggrell SA, Evans S (October 2003). “Treatment of dementia with neurotransmission modulation”. Expert Opinion on Investigational Drugs. 12 (10): 1633–54. doi:10.1517/13543784.12.10.1633. PMID 14519085. S2CID 46175609.
- ^ “Generally Recognized as Safe (GRAS) Determination for the Use of AlphaSize® Alpha-Glycerylphosphoryl Choline” (PDF). United States Food and Drug Administration. 25 January 2012. Archived from the original (PDF) on 24 December 2013.
- ^ Barbagallo Sangiorgi G, Barbagallo M, Giordano M, Meli M, Panzarasa R (June 1994). “alpha-Glycerophosphocholine in the mental recovery of cerebral ischemic attacks. An Italian multicenter clinical trial”. Annals of the New York Academy of Sciences. 717: 253–69. doi:10.1111/j.1749-6632.1994.tb12095.x. PMID 8030842. S2CID 86029937.
- ^ Traini, Enea; Bramanti, Vincenzo; Amenta, Francesco (December 2013). “Choline alphoscerate (alpha-glyceryl-phosphoryl-choline) an old choline- containing phospholipid with a still interesting profile as cognition enhancing agent”. Current Alzheimer Research. 10 (10): 1070–1079. doi:10.2174/15672050113106660173. ISSN 1875-5828. PMID 24156263.
- ^ Di Perri, R.; Coppola, G.; Ambrosio, L. A.; Grasso, A.; Puca, F. M.; Rizzo, M. (July 1991). “A multicentre trial to evaluate the efficacy and tolerability of alpha-glycerylphosphorylcholine versus cytosine diphosphocholine in patients with vascular dementia”. The Journal of International Medical Research. 19 (4): 330–341. doi:10.1177/030006059101900406. ISSN 0300-0605. PMID 1916007. S2CID 33715399.
- ^ Bellar, David; LeBlanc, Nina R.; Campbell, Brian (2015). “The effect of 6 days of alpha glycerylphosphorylcholine on isometric strength”. Journal of the International Society of Sports Nutrition. 12: 42. doi:10.1186/s12970-015-0103-x. ISSN 1550-2783. PMC 4650143. PMID 26582972.
- ^ Marcus, Lena; Soileau, Jason; Judge, Lawrence W.; Bellar, David (2017). “Evaluation of the effects of two doses of alpha glycerylphosphorylcholine on physical and psychomotor performance”. Journal of the International Society of Sports Nutrition. 14: 39. doi:10.1186/s12970-017-0196-5. ISSN 1550-2783. PMC 5629791. PMID 29042830.
- ^ Armah, Charlotte N.; Sharp, Paul; Mellon, Fred A.; Pariagh, Sandra; Lund, Elizabeth K.; Dainty, Jack R.; Teucher, Birgit; Fairweather-Tait, Susan J. (May 2008). “L-alpha-glycerophosphocholine contributes to meat’s enhancement of nonheme iron absorption”. The Journal of Nutrition. 138 (5): 873–877. doi:10.1093/jn/138.5.873. ISSN 1541-6100. PMID 18424594.